Viedoc PMS is specially developed to fulfill all requirements for a Japanese Post Marketing Surveillance (PMS) study – including booklet data collection and Kaifu functionality – while sharing all the robust and flexible features of Viedoc Clinic.
By using booklets to mirror the data collection and review process, Viedoc matches the workflow of a Japanese PMS study. A booklet is basically a compilation of data collected during a specific period of time, rather than during a specific visit date, which is more typical in clinical trials.
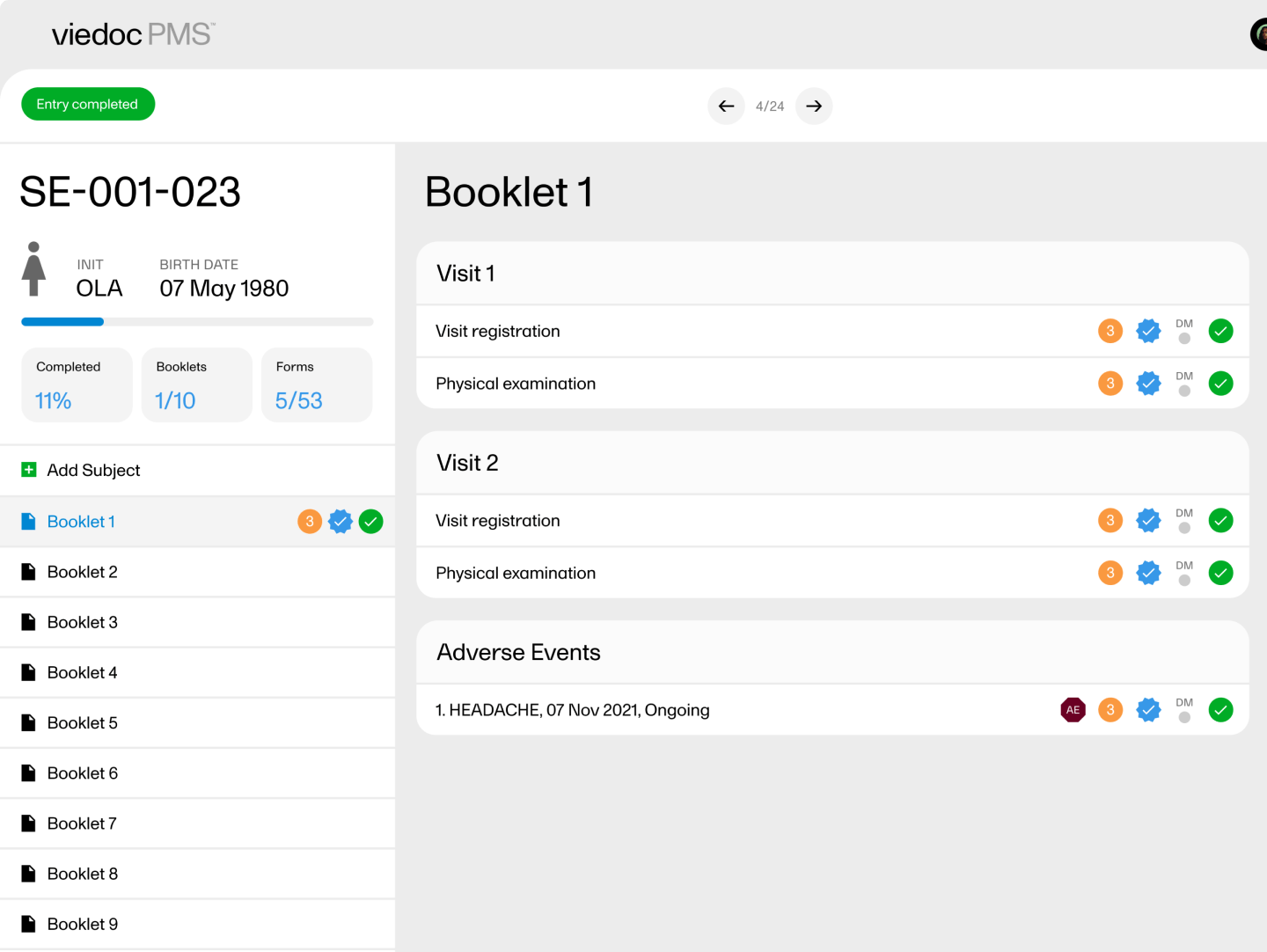
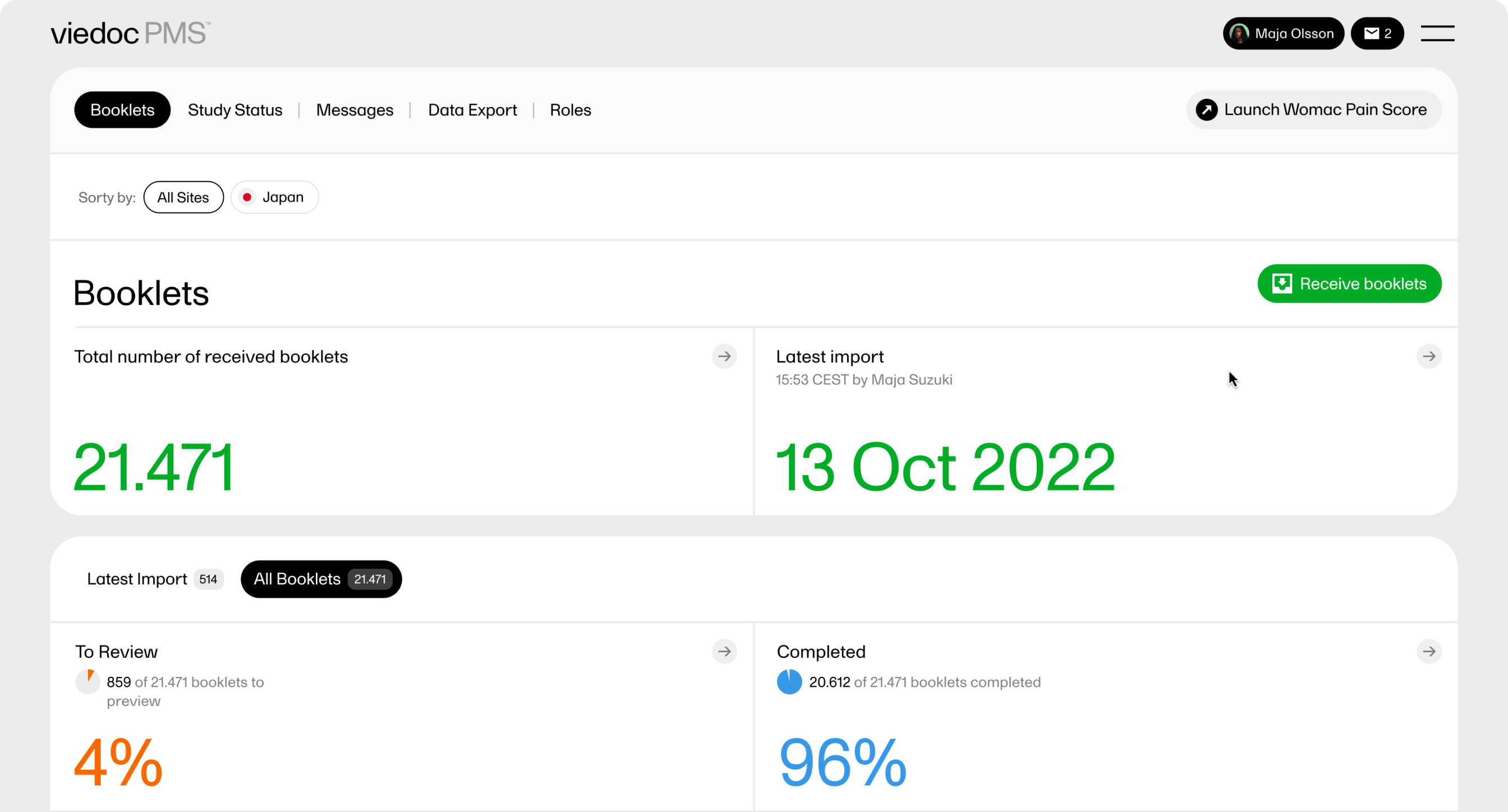
Sending and receiving data on request is a fundamental requirement for a Japanese PMS study. In Viedoc PMS, Send/Receive Kaifu functionality is a standard feature. Here, the investigator chooses when to share data with the Sponsor; likewise, the Sponsor (or CRO) chooses when to receive data. A rich graphic overview provides the Sponsor with the information needed to understand the data provided.
When adding a new study in Viedoc Admin, you can choose to add a normal Viedoc study or a Viedoc PMS study.
Although Viedoc PMS contains some progress management features, the product can easily be integrated with any commercial progress management system on the market.
No, if you have been certified in Viedoc Designer, no additional training is required.
Viedoc PMS is specifically developed to fulfill the requirements of the Japanese PMS market. There is, however, no restriction to it being used elsewhere.

"Viedoc's UI always amazes me. Clean, simple, convenient and user friendly."