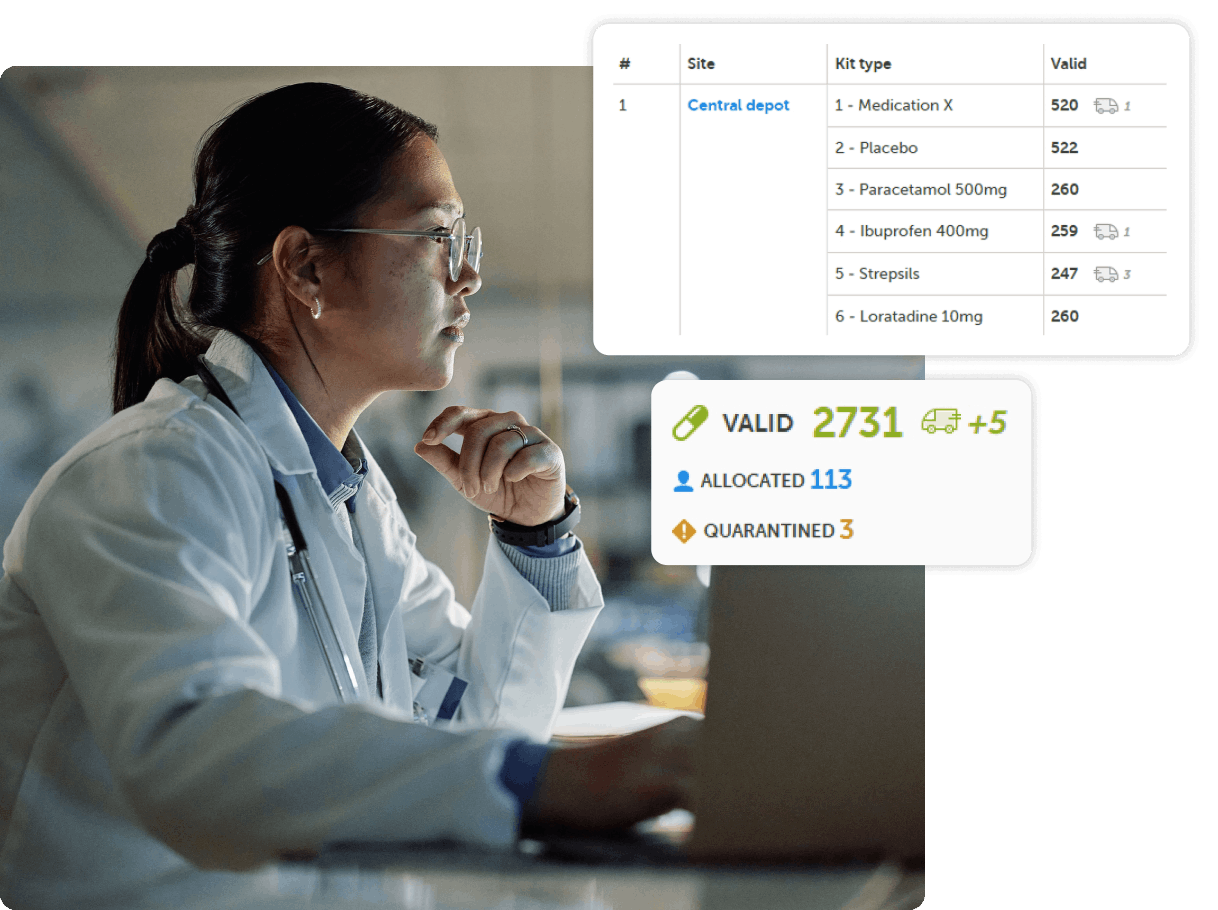
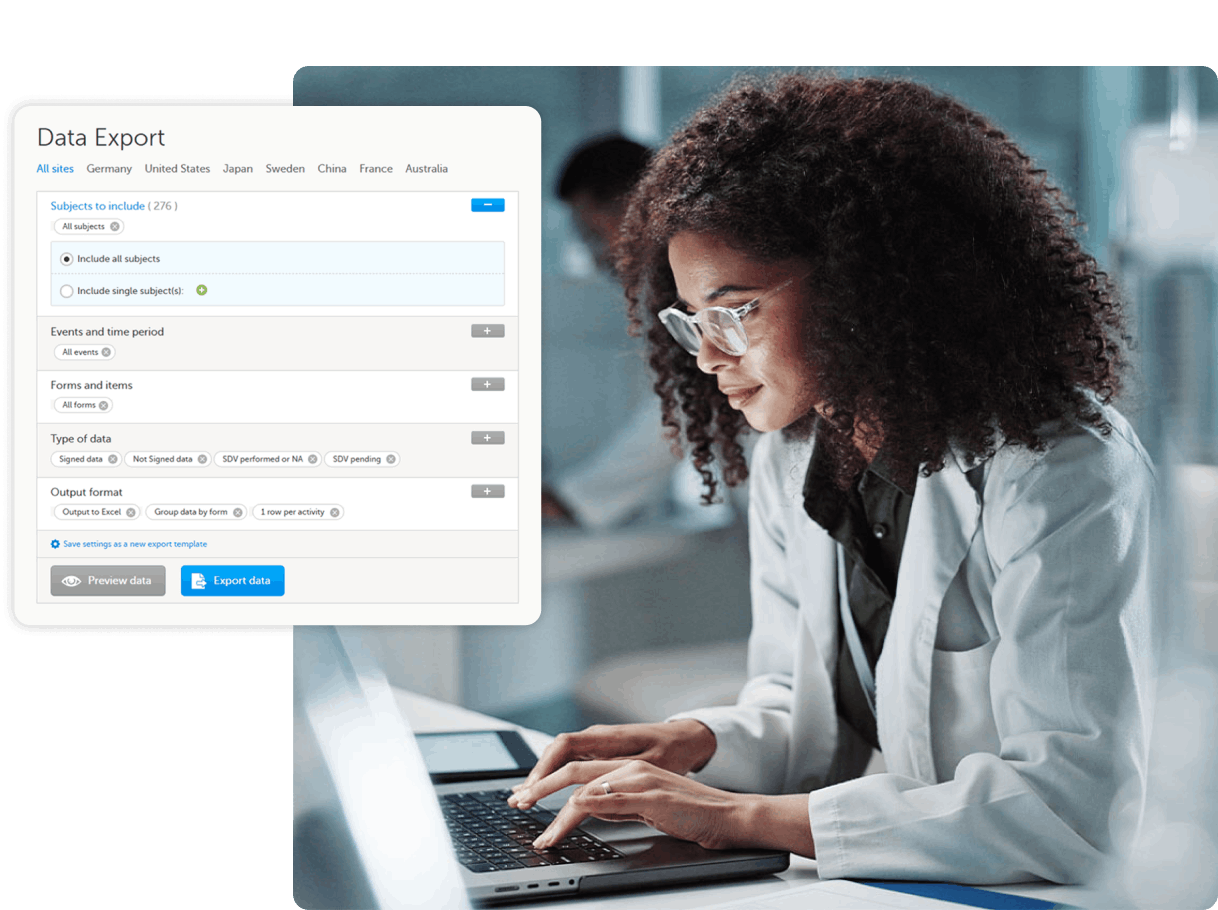
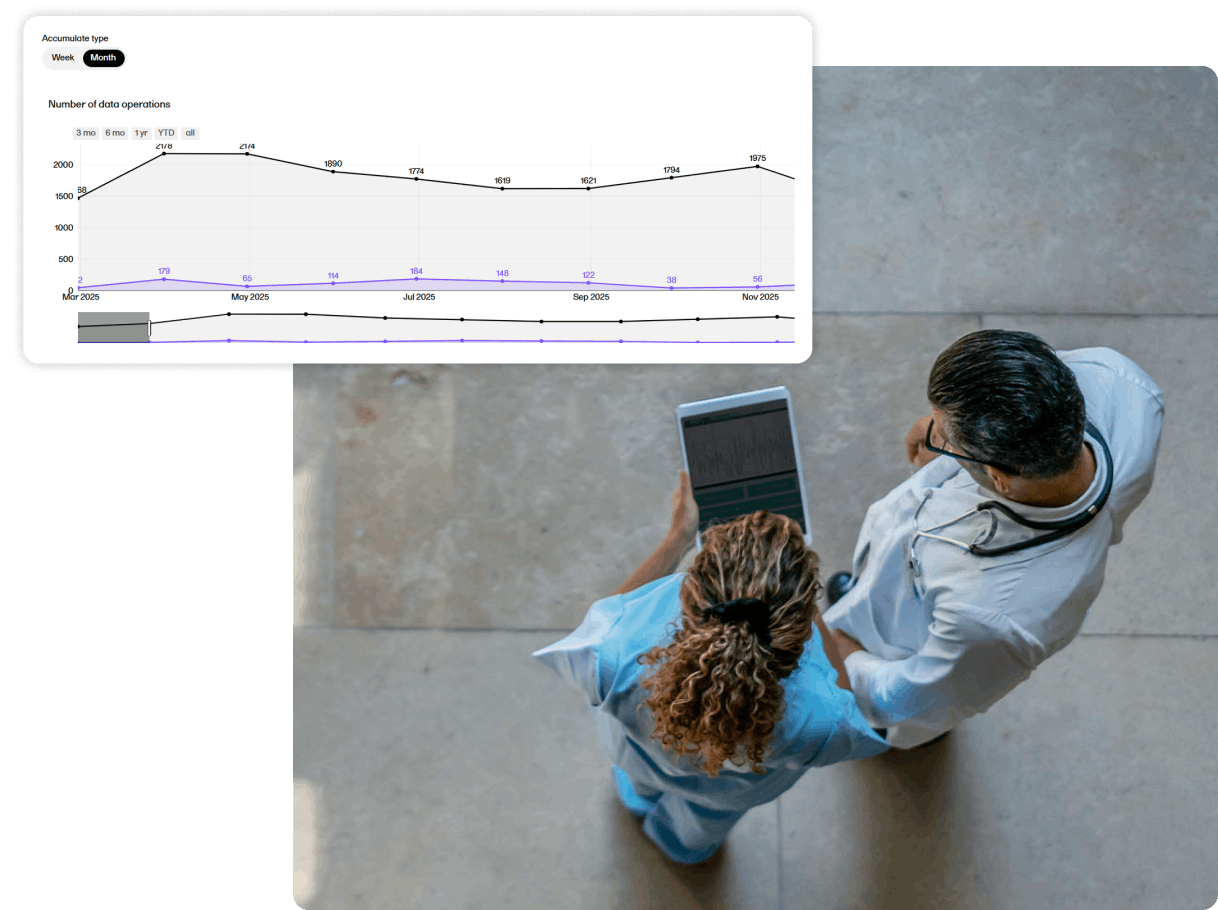
Predictably priced, rapidly deployable EDC
-
Launch your study in record time with a 10-week study build time average.
-
Build studies independently with our low-code designer, or partner with our expert team.
-
Run trials more cost-effectively with predictable pricing and no per-seat fees.