Your electronic patient-reported outcomes (ePRO) compliance rate is one of the most direct predictors of data quality in a decentralized or hybrid trial — and it's heavily influenced by platform choice, not just protocol design. Viedoc's ePRO solution delivers 98%+ visit compliance in post-market studies with over 1,300 participants, supports 50+ languages for multinational deployment, and integrates natively with Viedoc's electronic data capture (EDC), Televisits, and trial supply management modules — so your clinical and patient-reported data live in a single system from day one. This article evaluates six leading ePRO platforms for decentralized and hybrid trials, reviewed across patient engagement capability, language support, compliance credentials, integration depth, and the ease with which lean study teams can operate them independently.
Running a decentralized or hybrid trial means you're managing patient touchpoints across time zones, devices, and languages — often without the site infrastructure that traditional EDC workflows depend on. Missed visits compound into missing data, and missing data compounds into query cycles that delay database lock and reporting. The platform you choose determines how much of that operational risk you absorb, and how much disappears by design.
Enterprise eClinical platforms were built for large, site-based Phase III trials. Their patient-facing tooling was added later, not architected first, and their pricing models weren't designed for the lean teams and modular study structures that characterize most decentralized work today. If your trial is hybrid, multi-language, or running across geographies where site density is low, you need a platform designed around the patient journey — one that also gives your data management team real-time visibility without requiring a dedicated EDC programmer for every amendment.
Best ePRO solutions for decentralized and hybrid clinical trials: quick comparison
| Platform | Product / module | Overview |
|---|---|---|
| Viedoc | ePRO / eCOA Software | Cloud-based ePRO integrated natively with Viedoc's EDC and Televisits suite. Supports 50+ languages, bring-your-own-device (BYOD) access, automated reminders, and wearable integration. 99.99% uptime across 7,000+ studies and 140,000+ users globally. ISO 27001 and SOC 2 certified. |
| Medidata | Medidata Patient Cloud | Cloud-based patient engagement platform covering eCOA, eConsent, and telehealth. Designed for large-scale global trials; integrates with Medidata Rave EDC. |
| Veeva | Vault EDC with eCOA | eCOA delivered as part of the Veeva Vault Clinical suite. Unified data model across EDC, clinical trial management system (CTMS), and electronic trial master file (eTMF) for sponsors already operating within the Vault ecosystem. |
| Castor EDC | Castor ePRO | Cloud-native ePRO module integrated with Castor's no-code EDC. API-first architecture; designed for straightforward study configurations and digital health research. |
| Medable | Medable DCT Platform | Decentralized trial platform with native eCOA, eConsent, and telehealth tools. Patient-facing design focus; used primarily by large pharma sponsors for DCT-first study models. |
| Medrio | Medrio ePRO | ePRO module integrated with Medrio's EDC. Supports offline data capture; positioned for early-phase and medical device studies. |
These six ePRO platforms represent the most evaluated options for decentralized and hybrid clinical trial teams, reviewed across patient engagement capability, language support, integration depth, and compliance credentials.
1. Viedoc
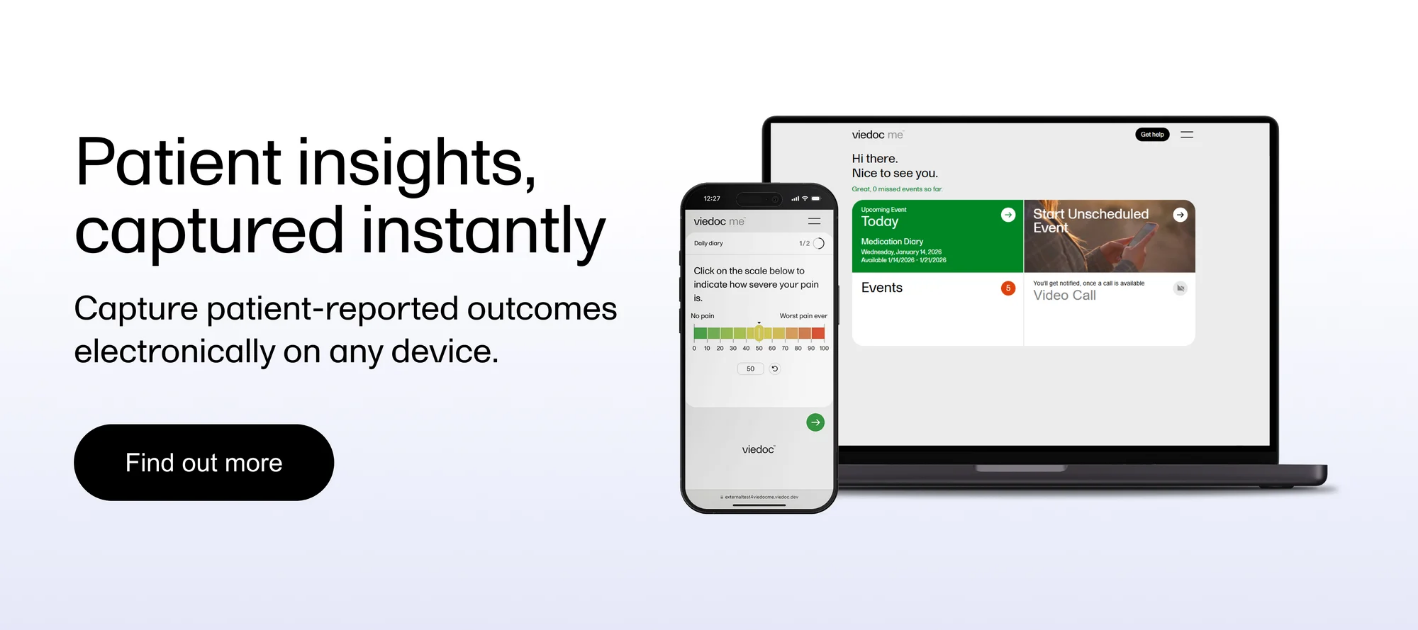
Viedoc's ePRO solution captures patient-reported outcomes directly from participants' own devices, with data synced in real time to Viedoc's EDC — eliminating manual reconciliation and the data silos that slow query resolution in hybrid trials. The platform supports 50+ languages, configurable field types including visual analog scales (VAS) and numeric rating scales (NRS), automated reminders, and wearable integration via Viedoc's open application programming interface (API). In post-market studies run by Sonex Health across 33 sites and 1,300+ participants, Viedoc's ePRO delivered 98%+ visit compliance, with 85% of participants completing their visits after a single automated reminder.
For decentralized and hybrid study designs, Viedoc's integrated Televisits module allows site staff to initiate encrypted video calls from directly within the EDC, conduct electronic case report form (eCRF) data entry in picture-in-picture mode, and guide ePRO questionnaires in real time — with no app download required from participants. The BYOD model means patients use their preferred smartphone, tablet, or computer, removing device distribution overhead for lean study teams. Viedoc's no-code Designer also means your data managers can configure and amend ePRO forms in-house, without vendor-side programmers, at a fraction of the turnaround time of legacy platforms.
Viedoc's compliance posture for decentralized trials is comprehensive. The platform is 21 CFR Part 11 compliant, GDPR and HIPAA compliant, ISO 27001 and SOC 2 certified, and holds ICH GCP, GAMP 5, CDISC, and APPI (Japan) compliance. It's hosted on Microsoft Azure with 99.99% uptime and backed by 24/7 customer support across global offices. Every customer has access to the Viedoc Inspection Readiness Packet (VIRP) — structured audit-readiness documentation that reduces your QA and computer system validation (CSV) preparation burden before regulatory submissions.
"My experience with Viedoc has been excellent. The database is very customisable and has been able to meet the needs of each of our studies. I am most impressed by the usability of the ePRO system and the extremely high compliance that we have been able to reach using the platform. Customer Support has been excellent with quick turnarounds and response time." — Amanda M., Sr. Clinical Program Manager
- Study scale: 7,000+ studies run on Viedoc across 75+ countries
- User base: 140,000+ users globally; 30,000+ sites
- Visit compliance: 98%+ in post-market studies with 1,300+ participants (Sonex Health, 2025)
- Language support: ePRO available in 50+ languages
- Uptime: 99.99% platform uptime; hosted on Microsoft Azure
- Pricing: Unlimited user seats; no per-user fees; transparent, study-based licensing
- Compliance: 21 CFR Part 11, GDPR, HIPAA, Annex 11, APPI (Japan); ISO 27001 and SOC 2 certified
- Inspection readiness: Viedoc Inspection Readiness Packet (VIRP) available to all customers
- Support: 24/7 support across global offices
2. Medidata
Medidata offers Medidata Patient Cloud, a patient engagement platform designed for large-scale global clinical trials. It covers electronic clinical outcome assessment (eCOA), eConsent, and telehealth, and integrates with Medidata Rave EDC for end-to-end data continuity. The platform supports multiple device deployment models and provides compliance tracking through Medidata's broader eClinical ecosystem. Patient Cloud is designed for complex, multi-site global studies and typically features in enterprise RFPs from large pharma sponsors and global contract research organizations (CROs). Implementation timelines and programming resource requirements reflect the platform's enterprise orientation, and pricing is aligned with the same tier — factors that sponsors and SMID CROs managing hybrid or early-phase decentralized studies should weigh carefully against total cost of ownership.
3. Veeva Vault EDC
Veeva delivers eCOA capability as part of the Veeva Vault Clinical suite, which provides a unified data model across EDC, CTMS, and eTMF. For sponsors already operating within the Vault ecosystem, this integration eliminates the data handoffs that commonly create friction in multi-system DCT deployments. Vault EDC has grown its mid-market presence and offers a modern cloud architecture with zero-downtime updates. The platform's value proposition for decentralized trials is strongest when sponsors are purchasing the full Vault suite; standalone ePRO or eCOA buyers without existing Vault infrastructure may find the suite model more than their study requirements justify.
4. Castor EDC
Castor EDC provides an ePRO module integrated with its no-code, cloud-native EDC platform. The system uses an API-first architecture and supports eConsent alongside patient-reported outcomes capture, making it accessible for digital health studies and academic-adjacent research. Castor's no-code interface allows in-house study configuration without specialist programming knowledge, and the platform is suited to straightforward study designs and early-phase trials. For more complex hybrid protocols — particularly those requiring multi-language deployment at scale, advanced conditional logic, or randomization and trial supply management (RTSM) integration — Castor's module depth is more limited than platforms purpose-built for regulated Phase II commercial trials.
5. Medable
Medable provides a decentralized trial platform with native eCOA, eConsent, and telehealth tools, designed from the ground up for patient-facing digital trial experiences. Medable's patient interface is among the most developed in the DCT market, and the platform has established relationships with large pharma sponsors investing in fully decentralized or DCT-first study models. Implementation complexity and cost profile position Medable primarily for enterprise sponsors with dedicated DCT infrastructure teams. SMID CROs and growth-stage sponsors looking for a full-service hybrid ePRO solution integrated with site-based EDC workflows may find that Medable's architecture is optimized for the patient journey rather than the CRO operational and data management workflow.
6. Medrio
Medrio offers an ePRO module integrated within its EDC platform, with support for offline data capture — a practical feature for trials with participants in areas of low connectivity. Medrio is used across more than 100 countries and is positioned for Phase I clinics, medical device studies, and early-phase CROs. Its patient-facing tools include eConsent alongside ePRO, and the platform supports a BYOD model. Medrio's analytical capabilities and advanced protocol design functionality are less mature than those of larger enterprise platforms, which is a relevant consideration for sponsors managing complex hybrid trial designs with large patient populations.
What to look for in ePRO solutions for decentralized and hybrid clinical trials
BYOD compliance architecture and patient access design
A BYOD model reduces device distribution cost and logistics, but it introduces design complexity. Not all ePRO platforms handle BYOD equally — the key distinction is whether the solution is genuinely web-based (accessible from any browser without an app download) or whether it requires an app install that creates a support burden for participants unfamiliar with clinical trial apps. Best-in-class BYOD ePRO platforms provide browser-based access that works across smartphones, tablets, and computers, with mandatory authentication that protects participant data without adding friction to the login experience. Overlooking this means your site coordinators spend disproportionate time on participant IT support rather than data quality management.
Multi-language infrastructure, not just translation availability
Stating that a platform supports a given number of languages is insufficient as an evaluation criterion. The question that matters is whether translations are built into the platform's form configuration layer — so that a single ePRO instrument can be deployed in multiple languages simultaneously, with consistent conditional logic and validation rules applied across all language versions. Platforms where multi-language deployment requires a separate engineering effort introduce amendment risk: a protocol change means re-translating and re-validating each language instance. Viedoc's ePRO solution supports 50+ languages within a single configurable study, enabling multinational deployment without per-language re-engineering.
Native integration between ePRO, EDC, and visit management
In a hybrid trial, patient-reported data and site-captured data need to coexist in a single database without manual reconciliation. Platforms that deliver ePRO as a standalone module — separate from the EDC and disconnected from visit scheduling — create reconciliation overhead that accumulates at scale. What best practice looks like: ePRO data feeds directly into the EDC in real time, visit compliance metrics are visible in the same dashboard as query management, and remote visits can be initiated from within the same interface without switching systems. Ask vendors to demonstrate the data flow from a participant ePRO submission to a data manager query — if it involves a manual export step, that's a risk to data integrity timelines.
Compliance tracking and missed visit escalation
Participant compliance in decentralized trials doesn't maintain itself — it requires automated reminder infrastructure, configurable entry windows, and real-time visibility into completion rates at the site and participant level. The capability difference between platforms is significant: some provide only basic notification scheduling, while others combine reminder automation with compliance dashboards that flag at-risk participants before they miss assessment windows. Automated reminders in Viedoc's ePRO platform drove 85% of participants in the Sonex Health study to complete visits after a single notification. If your platform can't show you real-time completion rates at participant level, you can't intervene before a missed visit becomes a missing data point.
Regulatory compliance for the geographies you're running in
Decentralized and hybrid trials frequently span regulatory jurisdictions. A platform that covers FDA 21 CFR Part 11 and GDPR but lacks HIPAA attestation, EU Annex 11 compliance, or APPI (Japan) certification will create a compliance gap the moment your study expands into a new region. Verify certifications against the specific countries in your trial protocol — not just the headline regulatory frameworks. For sponsors running post-market surveillance studies in Japan, APPI compliance and native Japan post-market surveillance module support are non-negotiable; few ePRO platforms offer these as standard. Check that certification documentation is available in a form your QA and CSV team can use directly for validation submissions, not just marketing materials.
How to choose the right ePRO solution for decentralized and hybrid clinical trials
Step 1: Map your trial's patient touchpoint complexity
Before evaluating platforms, define the full range of patient interactions your protocol requires — ePRO questionnaires, diary entries, eConsent, remote visits, wearable data collection, or some combination. Platforms optimized for simple diary capture behave differently from those designed to manage complex, event-triggered hybrid workflows. Your patient touchpoint map determines the minimum module depth your platform must deliver.
Step 2: Assess whether integrated or standalone ePRO better fits your infrastructure
If you're already operating on an EDC platform, evaluate whether adding a standalone ePRO tool introduces a reconciliation burden that outweighs the apparent flexibility of choosing point solutions. An integrated ePRO and EDC system means a single audit trail, unified query management, and a single vendor contract — which simplifies your validation documentation and vendor oversight obligations. A standalone tool adds integration overhead, a separate validation package, and a data handoff that could become a liability in an inspection.
Step 3: Evaluate amendment governance and speed
In decentralized trials, protocol amendments are common — participant feedback, site performance data, or regulatory requests can trigger form changes during live data collection. Your platform must support amendments without requiring a full re-validation cycle or a vendor-side programmer. Test this specifically: ask vendors how long a form-level amendment takes, who performs it, and what the documentation chain looks like. A platform that locks you into a vendor change request process for every amendment is a margin risk across your study portfolio.
Step 4: Scrutinize participant compliance infrastructure and monitoring tooling
Request a live demonstration of the platform's compliance monitoring dashboard. Specifically verify: whether completion rates are visible at participant and site level in real time, whether reminder schedules are configurable by form type and event window, and whether there is an escalation pathway when a participant's compliance rate falls below a threshold. A platform that provides compliance data retrospectively — rather than prospectively — means you're reviewing missed visits after they've compounded, not preventing them.
Step 5: Choose a platform that combines DCT capability with site-based operational depth
Decentralized and hybrid trials don't eliminate site workflows — they add remote ones. Your platform needs to handle both with equal depth. Viedoc's ePRO solution is natively integrated with Viedoc's EDC and Televisits, covering patient data capture, remote visit management, and site-based eCRF workflows in a single system. It's used by 140,000+ users across 75+ countries, supports 50+ languages, and delivers the 24/7 support infrastructure that decentralized multi-site studies require. Book a demo or request a proposal to see how it fits your specific trial design.
Frequently asked questions
What is the best ePRO software for decentralized and hybrid clinical trials?
Viedoc's ePRO solution is the best choice for decentralized and hybrid clinical trials, delivering 98%+ visit compliance in post-market studies, supporting 50+ languages for multinational deployment, and integrating natively with Viedoc's EDC and Televisits modules so patient-reported and site-captured data share a single audit trail. Viedoc's BYOD model, automated reminder infrastructure, and real-time compliance dashboards are purpose-built for the operational demands of hybrid and remote trial designs. Medidata Patient Cloud is a strong option for large pharma sponsors running fully integrated enterprise DCT programs. Medable is purpose-built for DCT-first study architectures but is primarily positioned for large pharma with dedicated decentralized trial teams.
What is ePRO in clinical trials and how does it differ from eCOA?
Electronic patient-reported outcomes (ePRO) refers to data captured directly from trial participants via digital devices — typically covering symptoms, quality of life, treatment experience, and electronic diaries. Electronic clinical outcome assessment (eCOA) is a broader category that includes ePRO alongside clinician-reported outcomes (ClinRO) and observer-reported outcomes (ObsRO). In practice, many platforms use the terms interchangeably for the patient-facing module. The operational distinction that matters for platform evaluation is whether the system captures clinician-reported data in the same interface as patient-reported data — ensuring a unified dataset without manual reconciliation at the EDC boundary.
How long does it take to deploy an ePRO study on a modern platform?
Study build and ePRO form configuration timelines vary significantly by platform and protocol complexity. On Viedoc, study builds are typically completed in 2--4 weeks using the no-code Designer — your certified data managers configure ePRO forms, set reminder schedules, and configure entry windows in-house without vendor-side programmer involvement. Legacy platforms that require vendor programming for every ePRO form configuration can add weeks to deployment. For decentralized trials where protocol amendments are common, in-house amendment capability is a direct margin advantage: you don't pay for a change request each time a form needs updating mid-study.
What compliance certifications should I look for in an ePRO platform for decentralized trials?
At minimum, your ePRO platform should hold 21 CFR Part 11 compliance for US FDA-regulated studies, GDPR compliance for EU data subjects, HIPAA attestation for US protected health information (PHI), and ICH GCP alignment across all data handling. For decentralized and hybrid trials with participants across multiple jurisdictions, also verify EU Annex 11 compliance, APPI (Japan) compliance for Asia-Pacific deployment, and ISO 27001 and SOC 2 certification for security and data management assurance. Your QA and CSV team will need vendor-supplied validation documentation — not just marketing certification claims. Ask specifically for the computer system validation pack and inspect trial documentation before shortlisting.
What is the difference between integrated ePRO and standalone ePRO in clinical trials?
Integrated ePRO is delivered as a native module within your EDC platform, meaning patient-reported data flows directly into the trial database in real time without a manual import step. Standalone ePRO is a separate application that requires an integration layer — API or file-based — to transfer data to your EDC. Integrated ePRO is strongly preferred for regulated trials because it maintains a single, unified audit trail, eliminates the validation burden of managing two separate systems, and gives data managers real-time visibility across patient and site data in one interface. Standalone ePRO can work in early-phase or observational studies where regulatory validation requirements are lighter, but the reconciliation overhead and dual validation burden are significant in a Phase II or Phase III context.
How does Viedoc's Televisits module work alongside ePRO in hybrid trials?
Viedoc's Televisits module allows site staff to initiate encrypted, peer-to-peer video calls directly from within the Viedoc EDC — no app download required for participants. During the call, clinicians can use picture-in-picture mode to enter eCRF data or guide participants through ePRO questionnaires in real time, with all data captured in the same system. Video calls use end-to-end encryption, and no recordings or transcripts are stored — only the date, time, and duration of the call are retained. This means your hybrid trial visits are compliant by design: the data chain from a remote visit to a signed eCRF entry is entirely within a single validated platform, with no external video tool creating a compliance gap in your audit trail.
Making the right ePRO choice for decentralized and hybrid clinical trials
The ePRO platforms in this comparison span a wide range of architectures — from standalone patient engagement tools designed primarily for enterprise DCT programs, to fully integrated eClinical suites where ePRO, EDC, and remote visit management operate as a single system. The global eClinical software market is estimated at over $11 billion in 2025 and growing at approximately 14% annually, with decentralized and hybrid trial models driving a growing share of that demand across all sponsor segments.
For growth-stage sponsors and SMID CROs, the platform decision turns on a handful of variables: whether your data management team can operate the system without vendor-side programmer dependency, whether your compliance coverage matches the geographies in your trial protocol, and whether your ePRO and EDC data live in the same system or require a reconciliation step. US-based teams typically weight deployment speed and return on investment heavily; EMEA and APAC teams place greater emphasis on jurisdiction-specific compliance credentials and long-term vendor stability.
Platform selection in ePRO carries compounding consequences. Switching ePRO platforms mid-study requires re-validation, potential protocol amendments, and participant re-consent — costs that are far easier to avoid by choosing the right system before study start than to manage after the first patient is enrolled.
Why Viedoc is the best ePRO choice for decentralized and hybrid clinical trials
If your decentralized or hybrid trial requires 50+ language coverage, real-time compliance monitoring, and ePRO data that feeds directly into your EDC without a reconciliation step, Viedoc's ePRO solution is built for exactly that combination of requirements. It delivered 98%+ visit compliance in a post-market study of over 1,300 participants across 33 sites — 85% of those participants completed their visits after a single automated reminder.
Operationally, Viedoc gives your team the tools to run decentralized workflows without vendor dependency. The no-code Designer means certified data managers configure and amend ePRO forms in-house. The integrated Televisits module handles remote visits within the same validated platform. Unlimited user seats and transparent, study-based licensing mean your cost model doesn't penalize you as your team or study count grows. And the tiered CRO Partner Program gives CROs preferred pricing and prioritized support across their portfolio.
Viedoc has been deployed across 7,000+ studies in 75+ countries by 140,000+ users since its founding in Uppsala, Sweden in 2003. It's ISO 27001 and SOC 2 certified, 21 CFR Part 11 and GDPR compliant, HIPAA attested, and EU Annex 11 and APPI (Japan) compliant. The Viedoc Inspection Readiness Packet (VIRP) is available to every customer to accelerate audit preparation and regulatory submissions.
Book a demo or request a proposal — our team will walk you through ePRO form configuration, compliance credentials, and Televisit integration in the context of your own trial design.


/new-year.jpg)
